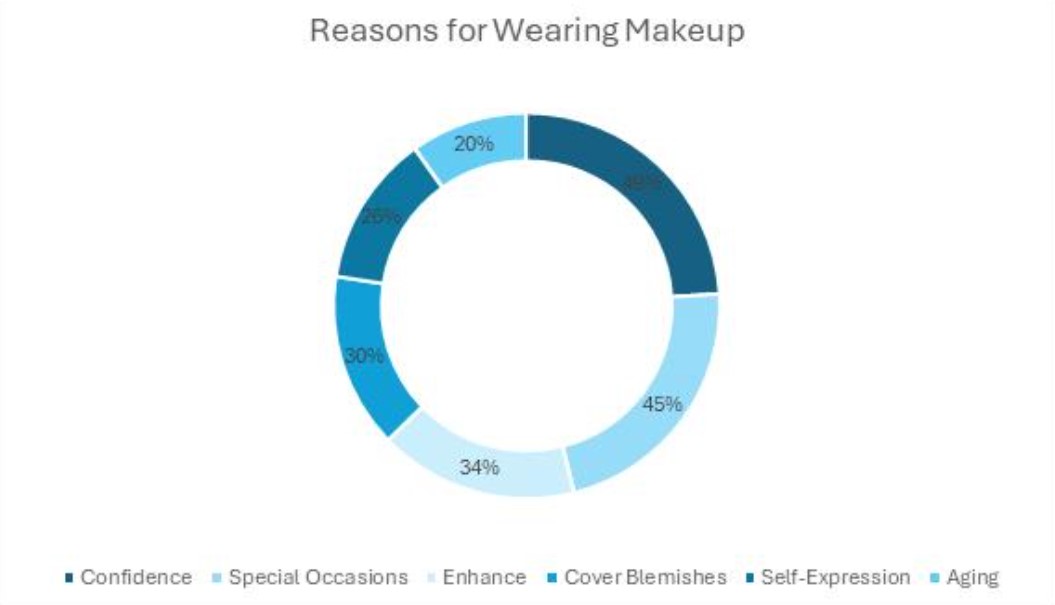
WASHINGTON – Acting Food and Drug Administration commissioner Andrew von Eschenbach testified last week that he decided non-prescription sales of the emergency contraceptive, Plan B, should be limited to women aged 18 and older even though the agency determined in 2005 that the drug could safely be sold over-the-counter to 17-year-olds.
Testifying at his nomination hearing before the Senate Health, Education, Labor and Pensions Committee, von Eschenbach said his decision was not based on new medical or scientific data or “political ideology” but rather his own “medical ideology” and his own interpretation of the scientific data.
After several years of delays, the Food and Drug Administration unexpectedly announced plans on Monday to make Plan B available without prescription to women ages 18 and older.
Barr Pharmaceuticals, the makers of Plan B, was seeking approval for non-prescription sales to women 16 and older.
Von Eschenbach testified that the decision to bar over-the-counter sales to women younger than 18 “was based primarily around our ability to manage” the sales of the drug, But he did not specify how.
He also said input from public-comment letters helped determine the new age limit for 18-year-olds, but again did not explain how.
The FDA did not respond to questions on these matters. Von Eschenbach did say that the age 18 cutoff provides “a greater safeguard in protecting and promoting the health” of young women, adding that it was similar to age restrictions on the sale of alcohol and tobacco to minors.
After reviewing previous studies about the safety of the drug, von Eschenbach said: “I’m of the opinion that the data is insufficient to be able to ensure safe and effective use of this drug by young women” under age 18 without medical supervision.
In response to speculation about outside political meddling in the Plan B case, von Eschenbach said: “No one told me what I could or couldn’t do. This was my assessment.”
But Sen. Patty Murray, D-Wash., provided an August 2005 letter from former FDA commissioner Lester Crawford that said agency researchers found the drug safe and effective for 17-year-olds.
The letter, to officials at a subsidiary of Barr Pharmaceuticals, said the FDA had “concluded the available scientific data are sufficient to support the safe use of Plan B as an over-the-counter product, but only for women who are 17 years of age and older.”