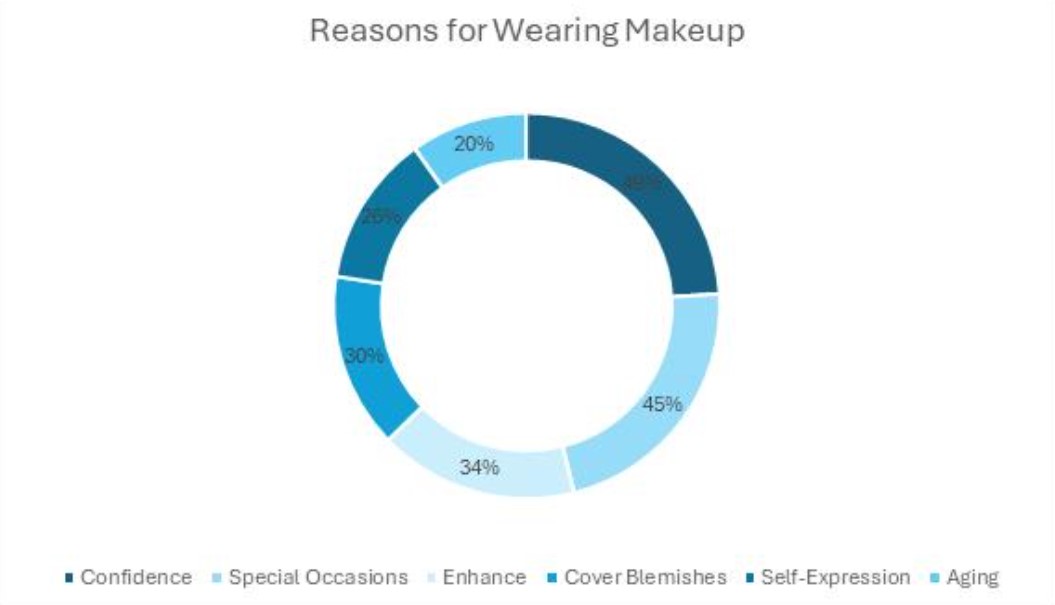
WASHINGTON – Federal health officials took unusual steps in rejecting nonprescription sales of emergency contraception – and some documents suggest the decision was made even before scientists finished reviewing the evidence, congressional investigators concluded.
An independent audit made public yesterday found the Food and Drug Administration’s May 2004 rejection of the politically charged morning-after pill deviated from 10 years of agency practice in switching drugs from prescription to over-the-counter sales.
Long-suspicious members of Congress immediately declared that politics had trumped science, and urged the FDA’s boss to intervene.
“We are deeply opposed to this subversion of science,” Rep. Henry Waxman, D-Calif., and 17 other lawmakers wrote Health and Human Services Secretary Mike Leavitt yesterday. “It appears that the decision … was preordained from the outset.”
Also, the lawmakers asked Leavitt to probe whether the FDA illegally destroyed documents from the office of then-Commissioner Mark McClellan, now the government’s Medicare chief, that might have shed more light on the controversial decision.
Leavitt’s office didn’t return phone calls seeking comment.
But in a statement, the FDA stood by its rejection and said the independent Government Accountability Office “mischaracterizes facts.”
Yesterday’s report is the latest blow to the credibility of an agency that by law is supposed to base decisions on science, not politics or industry pressure. Top-ranking FDA officials have acknowledged they overruled their own scientists’ decision that nonprescription sales of emergency birth control would be safe – and the agency’s women’s health chief resigned in protest.
A high dose of regular birth control, the morning-after pill lowers the risk of pregnancy by up to 89 percent if taken within 72 hours of unprotected sex. time.